VERONA Study Design:
Scheduled aflibercept
Scheduled visit
Sham injection
Protocol prespecified Retreatment criteria:
Starting at Week 4: BCVA reduction 5–9 letters and CST increase >75 µm on 2 consecutive visits†‡ OR
BCVA reduction ≥10 letters due to DME† OR
CST increase ≥100 µm‡ vs baseline OR
Investigator discretion
Starting at Week 12: Lack of 10% reduction in CST vs Baseline
Primary Endpoint:
Time to first supplemental anti-VEGF injection.
Patient Demographics:
Patients with active DME received ~2.7 anti-VEGF injections in the past year (mean; range 2–13 injections), diagnosed ~3 years prior (mean; range, 1–8 years), had a baseline of ~20/50 Snellen equivalent (mean 66–67 ETDRS letters; range 46–75 letters), and ~392–430 μm CST (mean; range, 320–645 µm).
*After screening, patients received a single aflibercept injection at Day 1 and were evaluated monthly for supplemental treatment (off-label aflibercept use). There was no loading phase.
†Relative to best on-study measurement.
‡CST as measured by SD-OCT.
VERONA Results
Primary Endpoint Met:
Both EYP-1901 dose levels demonstrated extended time to first supplemental treatment vs aflibercept—with the majority of EYP-1901 patients supplement-free up to Week 24
Cumulative supplement-free rates up to week 24, full analysis set§
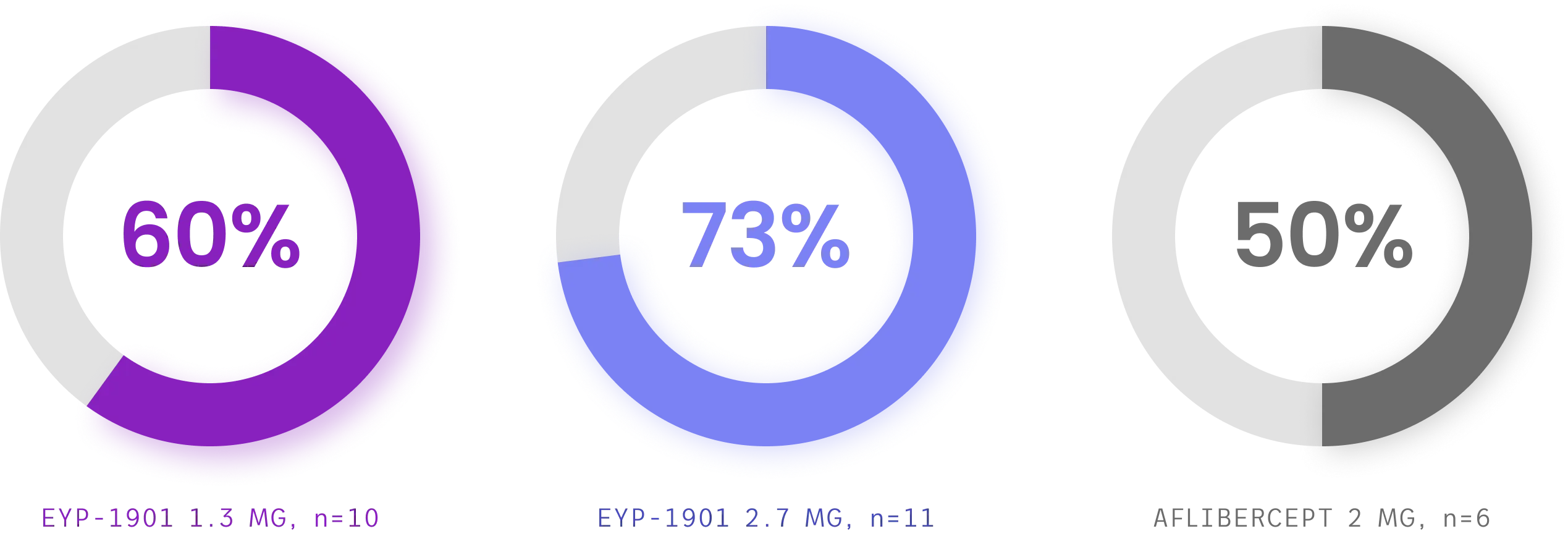
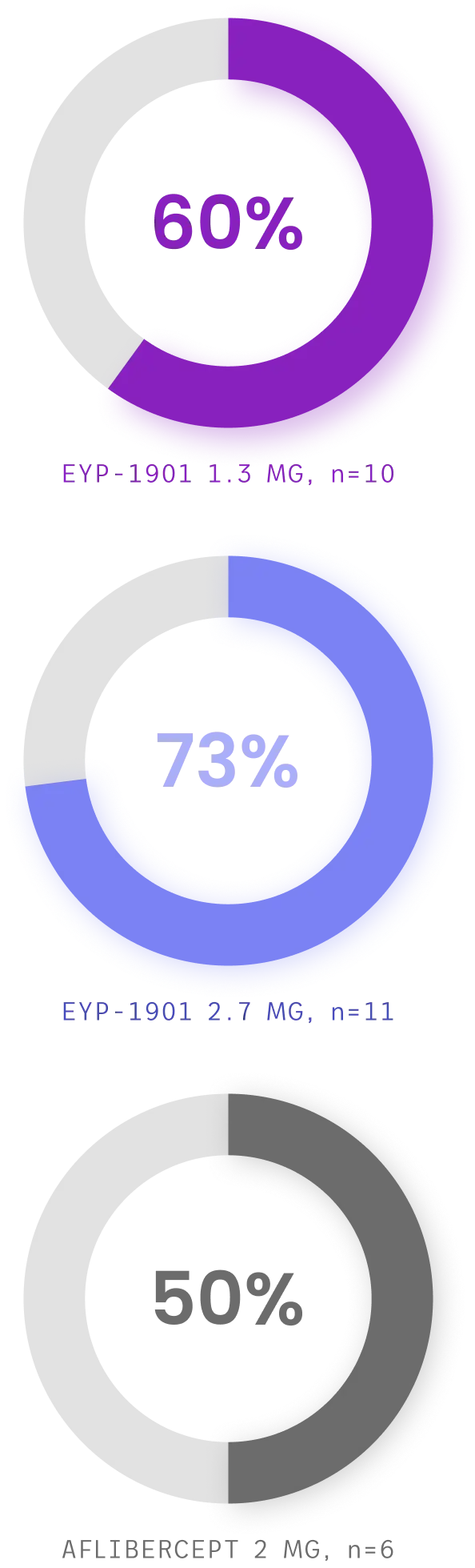
Both EYP-1901 dose levels demonstrated extended time to first supplemental treatment vs aflibercept
Summary of Eyes Requiring Supplemental Injections, Full Analysis Set
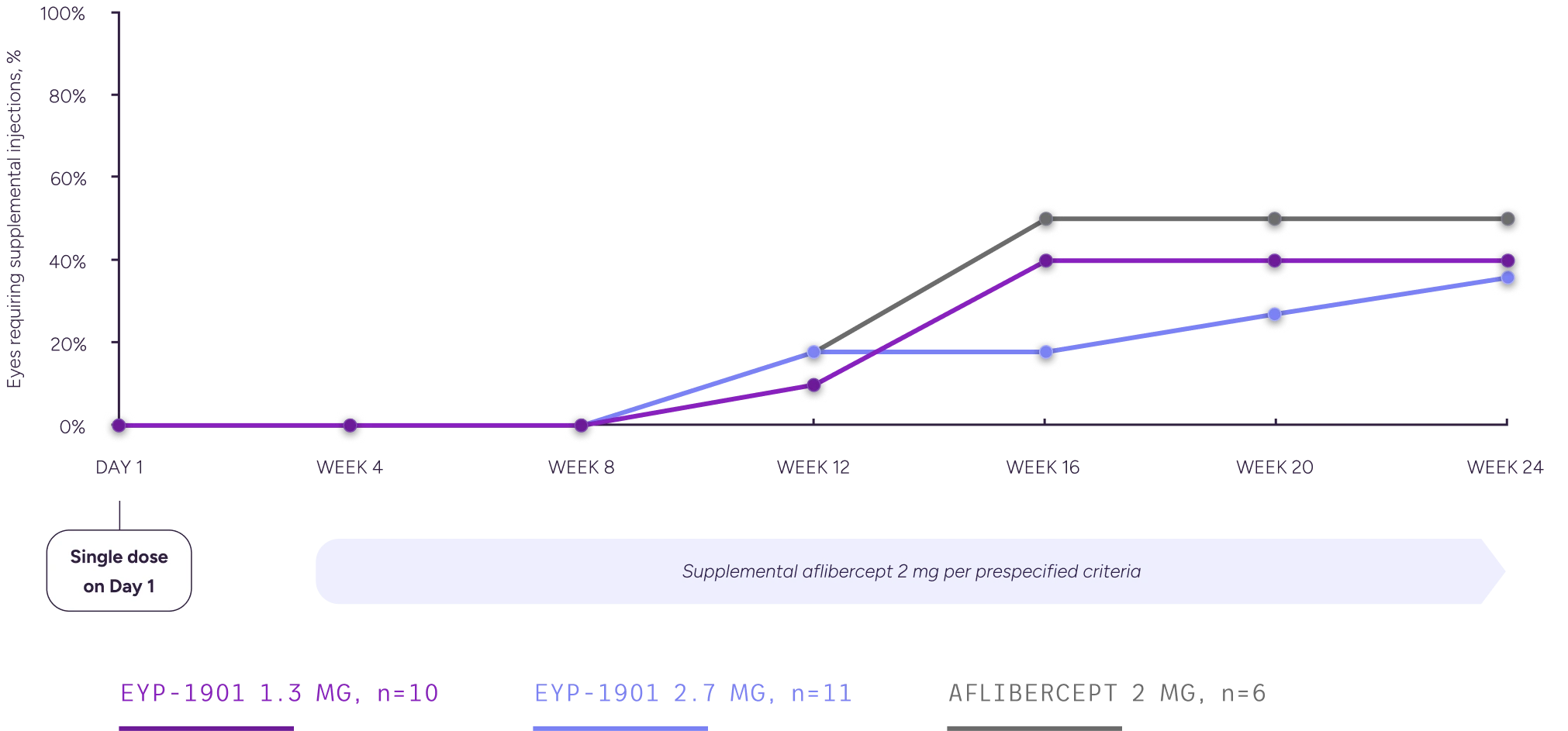
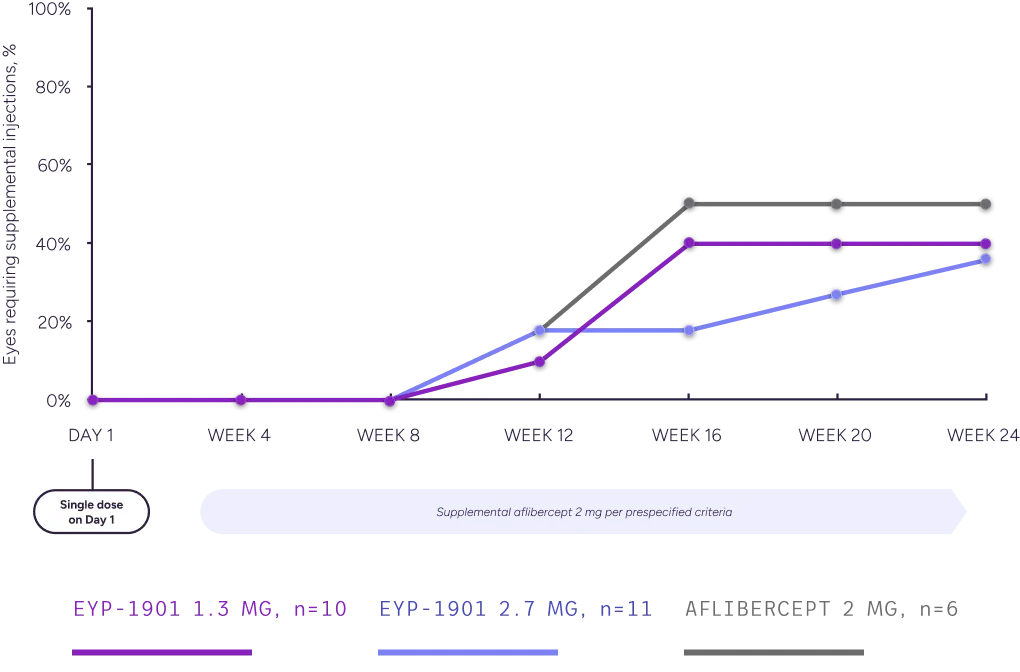
Graph depicts supplemental-injection rates through (ie, including) the specified study week.
§Supplement-free rates up to Week 24 reflect rates prior to any treatments given during the Week 24 visit.
BCVA and CST outcomes: A single EYP-1901 dose resulted in a clinically meaningful improvement with early bioavailability
BCVA and CST Changes From Baseline, Full Analysis Set
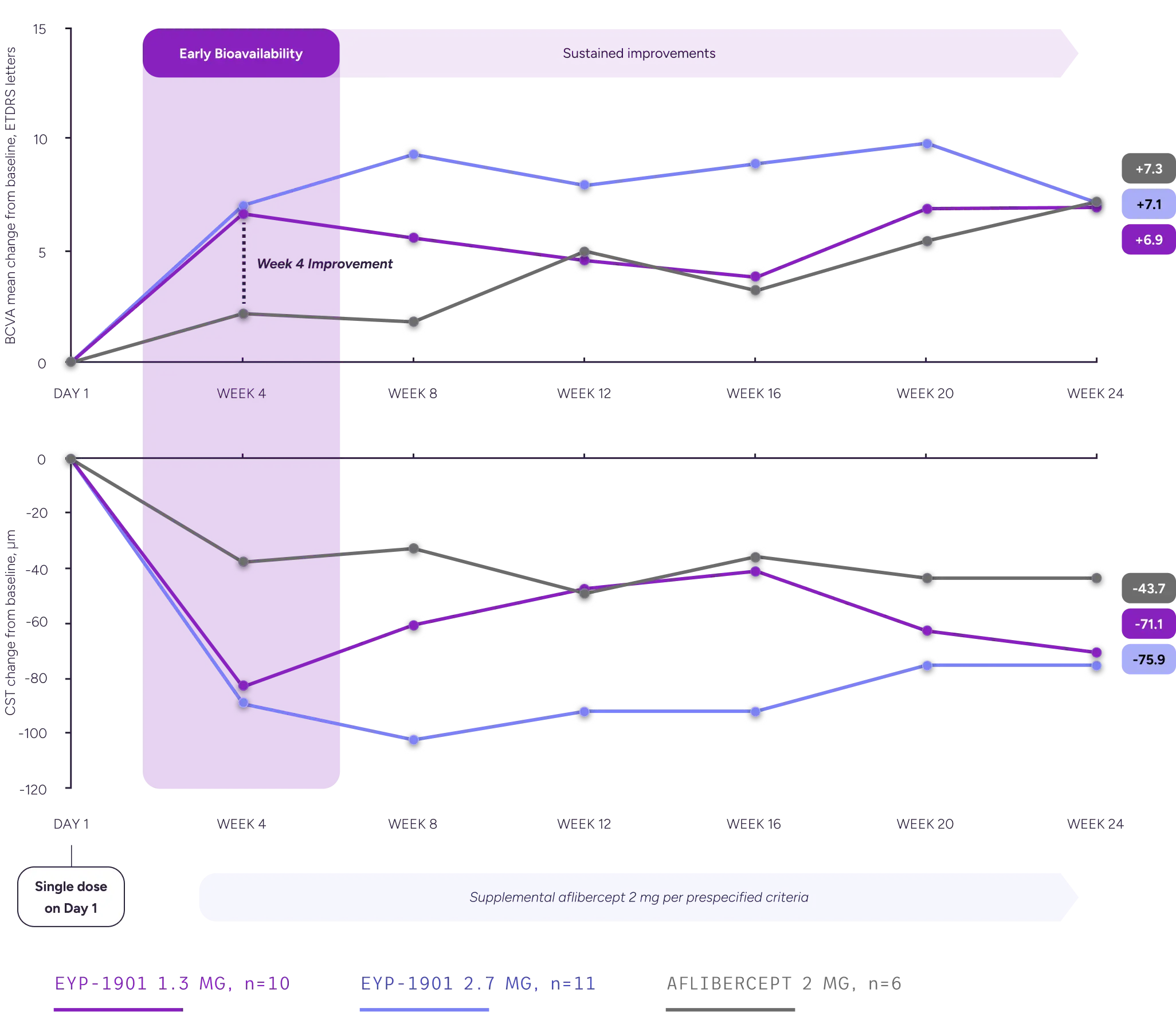
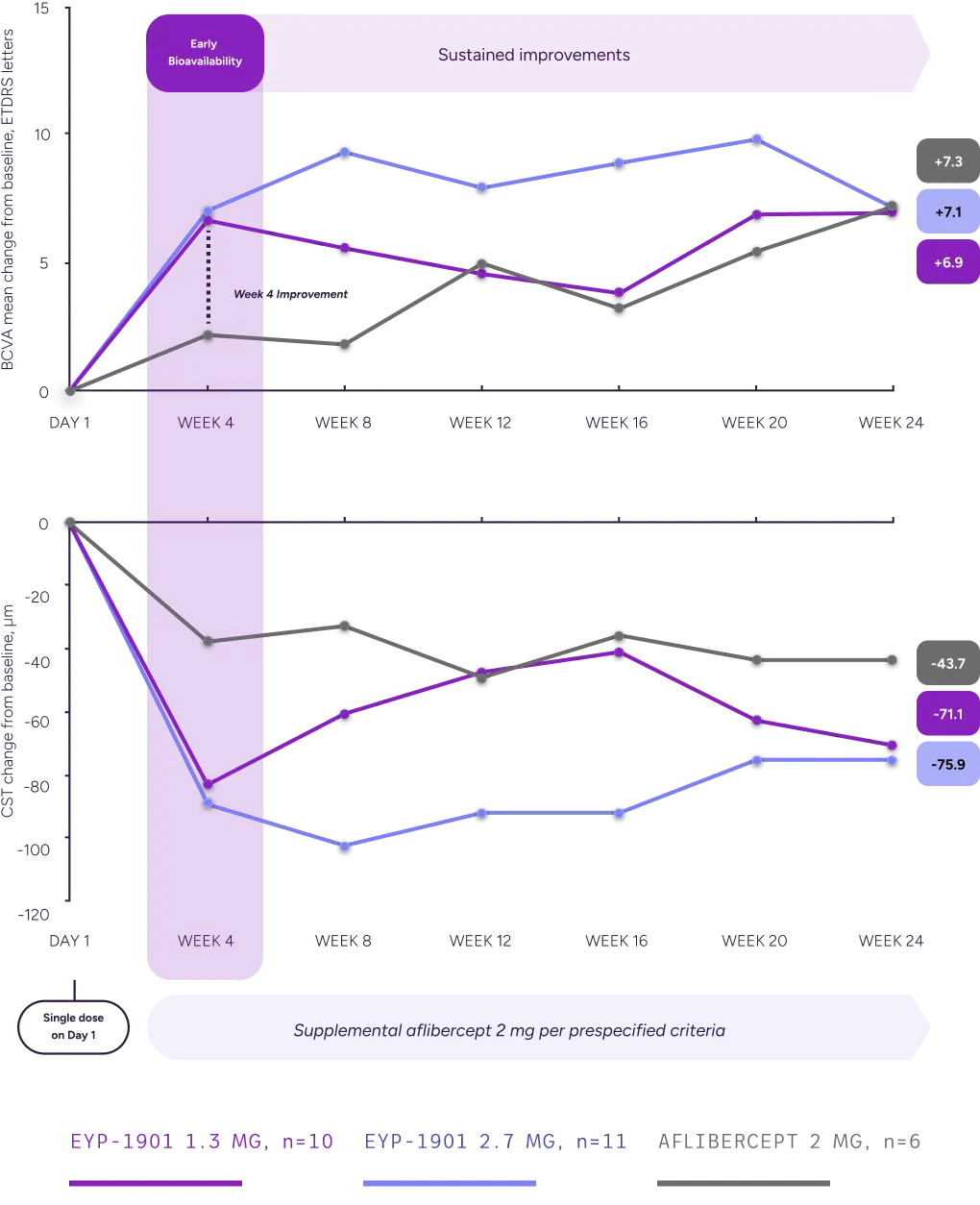
In a subgroup analysis of supplement-free eyes, a single dose of EYP-1901 demonstrated better BCVA and fluid outcomes vs aflibercept in eyes that remained supplement-free through Week 24
BCVA and CST Changes From Baseline, Supplement-Free Subgroup
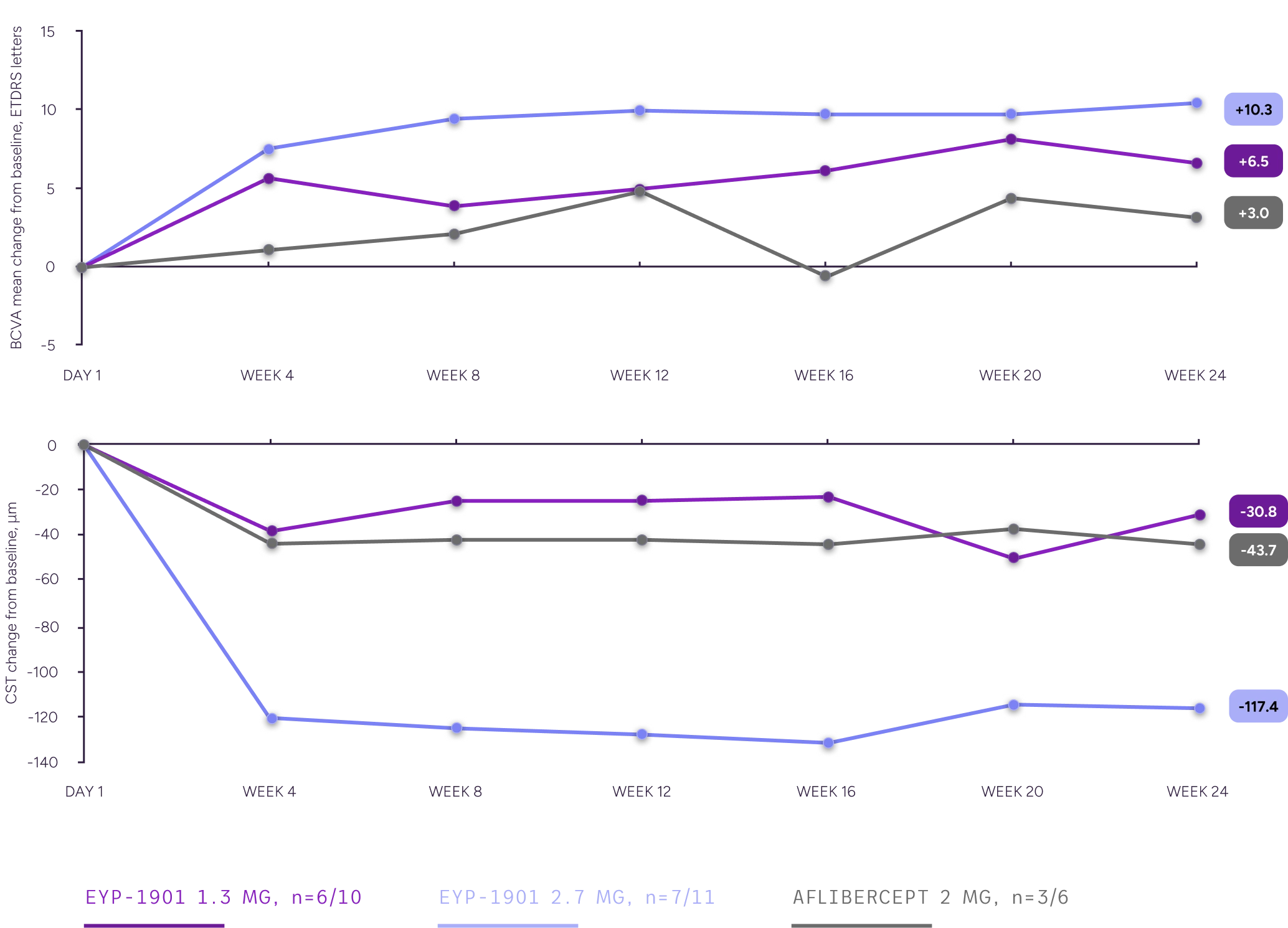
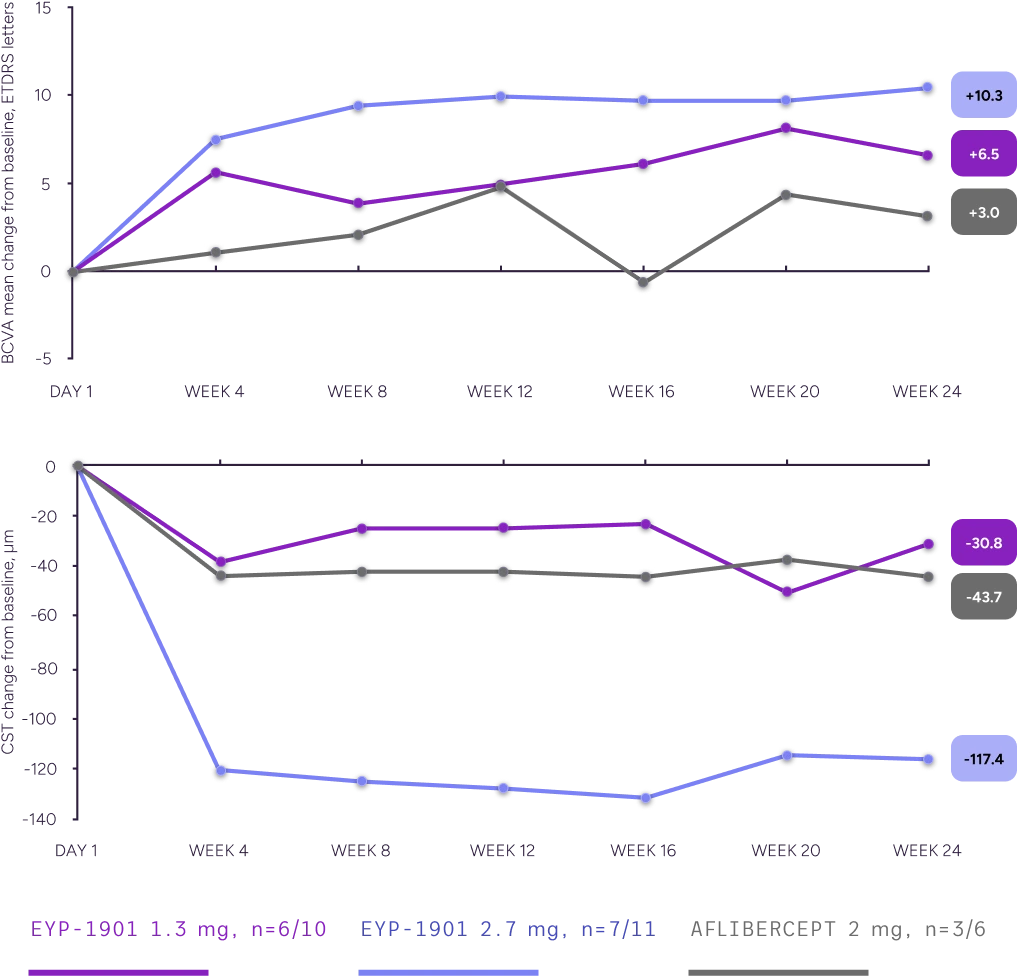
Supplement-free is defined as patients who did not receive a supplement at any point during the study.
A Favorable Safety Profile
EYP-1901 was well-tolerated—No SAEs related to EYP-1901 reported
VERONA Key Safety Findings
- No EYP-1901-related ocular or systemic SAEs
- No cases of:
- Endophthalmitis
- Retinal vasculitis (occlusive or non-occlusive)
- Intraocular inflammation (IOI)
- Insert migration into the anterior chamber
- No visual impairment due to the insert
- No discontinuations
BCVA, best corrected visual acuity; CST, central subfield thickness; DME, diabetic macular edema; ETDRS, Early Treatment Diabetic Retinopathy Study; SAE, serious adverse event; SD-OCT, spectral-domain optical coherence tomography; VEGF, vascular endothelial growth factor.

